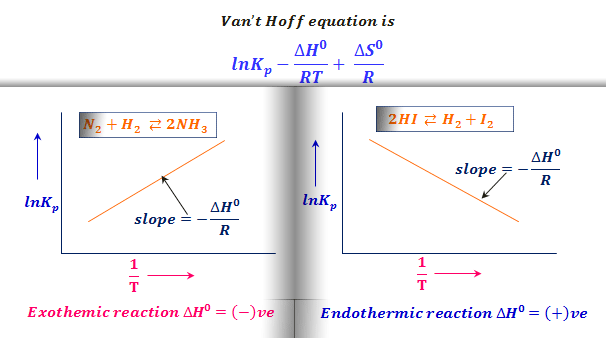
Jacobus Henricus van 't Hoff - Integrated van't Hoff equation (if the standard reaction enthalpy ΔH0 is known and constant in the temperature range of the measurement) is used for temperature correction.

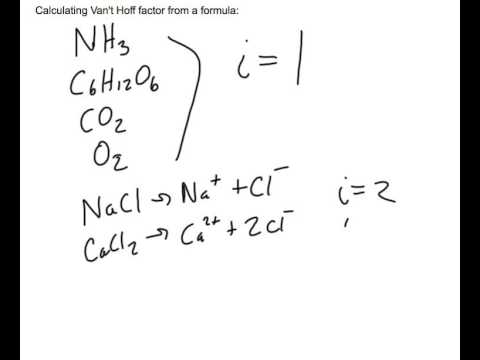
SOLVED: What is the predicted van't Hoff factor (i) for each of the following compounds? Enter only a number as an answer for each, such as 0, 1, 2, ... CH4 AlCl3

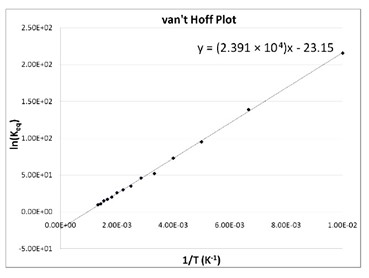
Van't Hoff plot of ln K H vs. inverse temperature for adsorption of... | Download Scientific Diagram